FDA Grants Conditional Approval To Novavax COVID-19 Vaccine: What You Need To Know
Welcome to your ultimate source for breaking news, trending updates, and in-depth stories from around the world. Whether it's politics, technology, entertainment, sports, or lifestyle, we bring you real-time updates that keep you informed and ahead of the curve.
Our team works tirelessly to ensure you never miss a moment. From the latest developments in global events to the most talked-about topics on social media, our news platform is designed to deliver accurate and timely information, all in one place.
Stay in the know and join thousands of readers who trust us for reliable, up-to-date content. Explore our expertly curated articles and dive deeper into the stories that matter to you. Visit Best Website now and be part of the conversation. Don't miss out on the headlines that shape our world!
Table of Contents
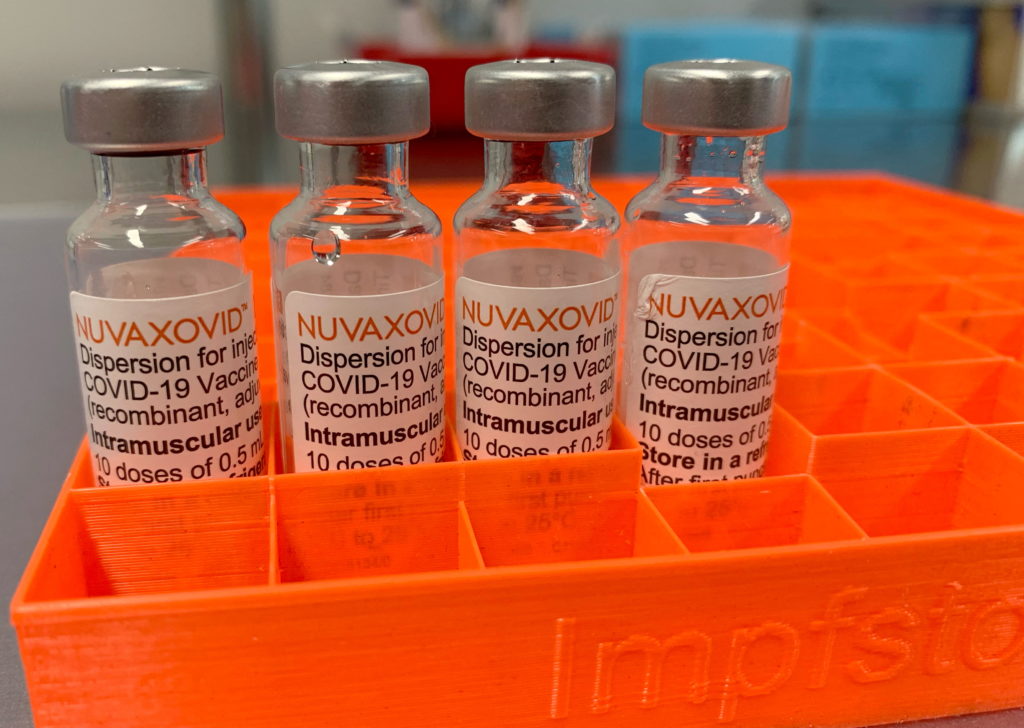
FDA Grants Conditional Approval to Novavax COVID-19 Vaccine: What You Need to Know
The United States Food and Drug Administration (FDA) has granted conditional approval to Novavax's COVID-19 vaccine, Nuvaxovid, marking a significant development in the ongoing fight against the pandemic. This approval offers a new option for individuals seeking COVID-19 vaccination, particularly those who may have hesitated due to concerns about mRNA technology used in other vaccines. This article will delve into the key details surrounding this approval and what it means for you.
What is Nuvaxovid?
Nuvaxovid is a protein subunit vaccine, a different technology compared to the mRNA vaccines from Pfizer-BioNTech and Moderna. Instead of using mRNA to instruct cells to produce a viral protein, Nuvaxovid uses a lab-made version of the spike protein found on the surface of the SARS-CoV-2 virus. This spike protein is then combined with an adjuvant, a substance that enhances the immune response. This approach has been used in other successful vaccines, such as those for Hepatitis B and the flu.
Why is this Approval Significant?
The approval of Nuvaxovid offers several key advantages:
- Alternative Technology: For individuals hesitant about mRNA vaccines, Nuvaxovid provides a different technological approach, potentially increasing vaccine uptake.
- Increased Vaccine Supply: This approval adds to the overall supply of COVID-19 vaccines available, enhancing the capacity to vaccinate more people globally.
- Potential for Broader Use: Its different mechanism may prove effective against emerging COVID-19 variants. Further research is needed to confirm this.
What are the potential benefits and risks?
Like all vaccines, Nuvaxovid carries potential benefits and risks. The benefits include protection against severe illness, hospitalization, and death from COVID-19. Common side effects reported during clinical trials include pain at the injection site, fatigue, headache, muscle pain, and joint pain. These side effects are generally mild and temporary. Serious side effects are rare. You should always discuss any concerns with your healthcare provider before getting vaccinated.
Who Should Get the Novavax Vaccine?
The FDA's authorization makes Nuvaxovid available to individuals 18 years of age and older. Consult your doctor to determine if this vaccine is the right choice for you based on your individual health history and risk factors.
What does Conditional Approval Mean?
Conditional approval means the FDA has granted approval based on promising clinical trial data, but the agency will continue to monitor the vaccine's safety and effectiveness in the real world. Novavax is required to submit additional data to the FDA to confirm the long-term safety and efficacy of the vaccine.
Where Can I Get the Novavax Vaccine?
The availability of Nuvaxovid will vary by location. Check with your local health department or healthcare provider to find out where you can get vaccinated. You can also visit the CDC website ([link to CDC website]) for updated information on vaccine availability.
Conclusion:
The FDA's conditional approval of the Novavax COVID-19 vaccine is a welcome development. This new vaccine offers an alternative approach to vaccination and could help increase overall vaccination rates. Remember to consult your doctor to discuss whether the Novavax vaccine is the right choice for you and stay informed about the latest developments in the fight against COVID-19. Getting vaccinated remains a crucial step in protecting yourself and your community.

Thank you for visiting our website, your trusted source for the latest updates and in-depth coverage on FDA Grants Conditional Approval To Novavax COVID-19 Vaccine: What You Need To Know. We're committed to keeping you informed with timely and accurate information to meet your curiosity and needs.
If you have any questions, suggestions, or feedback, we'd love to hear from you. Your insights are valuable to us and help us improve to serve you better. Feel free to reach out through our contact page.
Don't forget to bookmark our website and check back regularly for the latest headlines and trending topics. See you next time, and thank you for being part of our growing community!
Featured Posts
-
 New Art Installation At Guaranteed Rate Field A White Sox Homage To Pope Leo Xiii
May 21, 2025
New Art Installation At Guaranteed Rate Field A White Sox Homage To Pope Leo Xiii
May 21, 2025 -
 Fbi Probe Of New York Attorney General Confirmed What We Know
May 21, 2025
Fbi Probe Of New York Attorney General Confirmed What We Know
May 21, 2025 -
 Nfl Owners To Tackle Controversial Tush Push Ban And Future Of The Game
May 21, 2025
Nfl Owners To Tackle Controversial Tush Push Ban And Future Of The Game
May 21, 2025 -
 Marvin Harrison Jr Bulks Up Can Added Muscle Translate To On Field Success For The Buckeyes
May 21, 2025
Marvin Harrison Jr Bulks Up Can Added Muscle Translate To On Field Success For The Buckeyes
May 21, 2025 -
 Update On Christen Press Angel City Forward Released Following Medical Emergency
May 21, 2025
Update On Christen Press Angel City Forward Released Following Medical Emergency
May 21, 2025
